
Case Study Biodimed
Support for a Biotech Company entering the french market with an orphan drug.
Support for a Biotech Company entering the french market with an orphan drug.

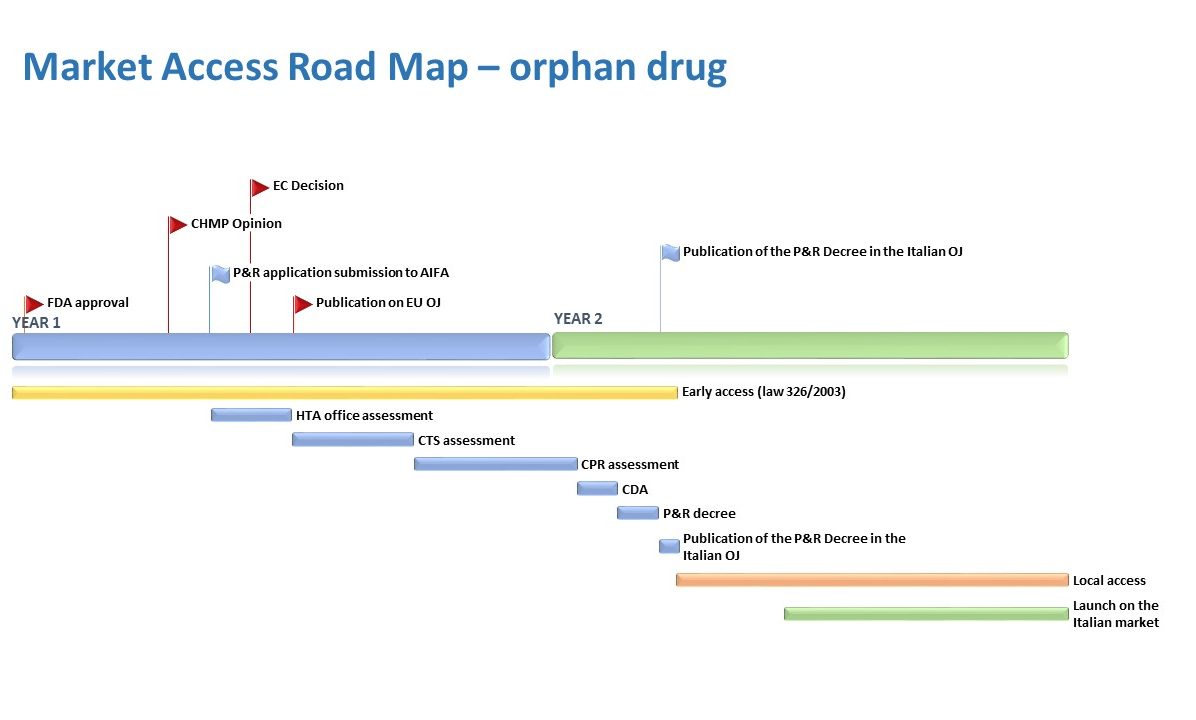
RPN has experience with a wide range of therapeutic areas and particularly with orphan and advanced-therapy medicinal products. RPN support includes early access and P&MA strategic assessments, advisory boards, stakeholder management and full support with national and regional P&R negotiations.
In order to develop a complete and successful access plan, RPN proposes specific strategic approaches that accelerate the accessibility of the therapeutic solution to meet patient clinical needs.
The initial approach generally involves the analysis of current therapeutic algorithm in which the product should be positioned and analysis of strengths/hurdles. In parallel, data can be shared and discussed with KOLs and Payers to support the definition of strategy, clarify the precise place in therapy of the new technology and the specific unmet need recognized at national level.
After defining these first aspects, RPN can develop, in advance to many countries, an effective EAP (early access program) that allows rapid availability of the new drug for the benefit of patients.
At the same time, in the pre-P&R submission phase, RPN can support with the development of HE Models which identify and highlights the economic/social impact that the new therapeutic solution can have on the Italian environment.
On the basis of the strategic analysis performed and the wide experience in the field, RPN can support with the preparation of the Pricing & Reimbursement Dossier and the Innovation application. After the submission to AIFA, RPN supports clients with close follow-up and attendance of meetings with AIFA Commissions up to the publication of the P&R decree in the Italian Official Journal.
Finally, RPN can support and coordinate the client’s regional activities in such a way as to accelerate and optimize the timing of regional access in the Italian Local Health Units.

Support for a Biotech Company entering the french market with an orphan drug.



We are pioneers in the utilisation of Multi-Criteria Decision Analysis (MCDA) at international and national level, helping evaluation committees to assess the relative additional value contribution of new health technology and helping companies to better position their product with respect to existing alternatives.

SmartStep Healthcare Strategy & Market Access Consulting provides personalized and specialized consulting services to the pharmaceutical and medical device industries to provide fast and effective market access to their products in Germany.