French HTA assessment in cases of significant uncertainty: Added value and reassessment
The TC Doctrine provides that “Pending new data, a sufficient CB may be granted on a conditional and exceptional basis in situations where the lack of reimbursement given the preliminary data is likely to result in a loss of opportunity for patients”:
- serious diseases, irrespective of its prevalence, and
- unmet medical need (not covered or insufficiently covered), and
- initial data suggestive of a clinical utility for the patient, and
- development plan enabling the elimination of uncertainties in the short term,
The reassessment of the CB will be considered in the light of the results submitted with a view to resolving the uncertainties […] and within a timetable defined by the Commission”. The TC Doctrine does not provide indication regarding the clinical added value level (CAV, Amélioration du Service Médical Rendu or ASMR) in these situations.
An analysis has been made of the TC opinions published up to October 2023.
Overall, 21 TC opinions on 22 indications were found, resulting in a total of 22 conditional CB assignments, the majority (62%) in oncology or haematology. Of these, 17 (76%) had an early access authorization in France and 13 (62%) had a conditional European marketing authorization (MA). Most of the CB cases were classified as mild (45%) or important (41%), with CAV V (86%). The TC requested additional data within a time frame ranging from one month (COVID) to five years (based on the conditional MAA timeline).
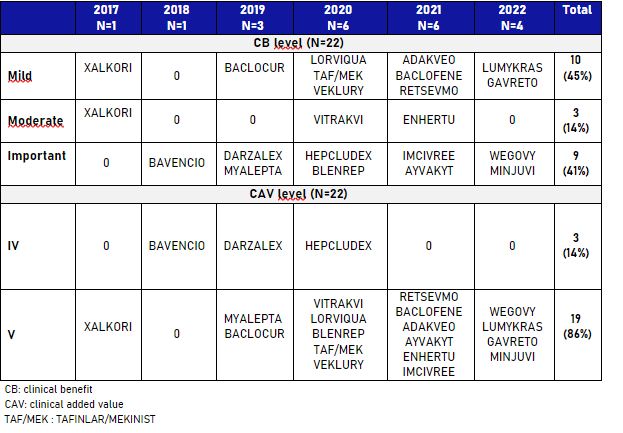
Nine (43%) have been reassessed based on the additional data and the reimbursement has been confirmed for all of them:
- 3 CAV were upgraded: IMCIVREE and VITRAKVI from CAV 5 to 4, and DARZALEX from CAV 4 to 3
- 2 CAV were downgraded: XALKORI and BAVENCIO from CAV 4 to 5
- 4 CAV were maintained: RETSEVMO, LUMYKRAS, LORVIQUA, TAFINLAR/MEKINIST
This ‘conditional CB’, implemented by the TC in its 2018 Doctrine, has provided or maintained access to 21 medicinal products, in a situation of early data packages, with the aim of supporting access to innovation for patients.
As the PHAROS Group, we are well equipped to provide expert guidance and support for French HTA assessments in cases of significant uncertainty, with a particular focus on added value and reassessment.