How to anticipate the 2025 EU HTA from now?
The future scope of the EU assessment
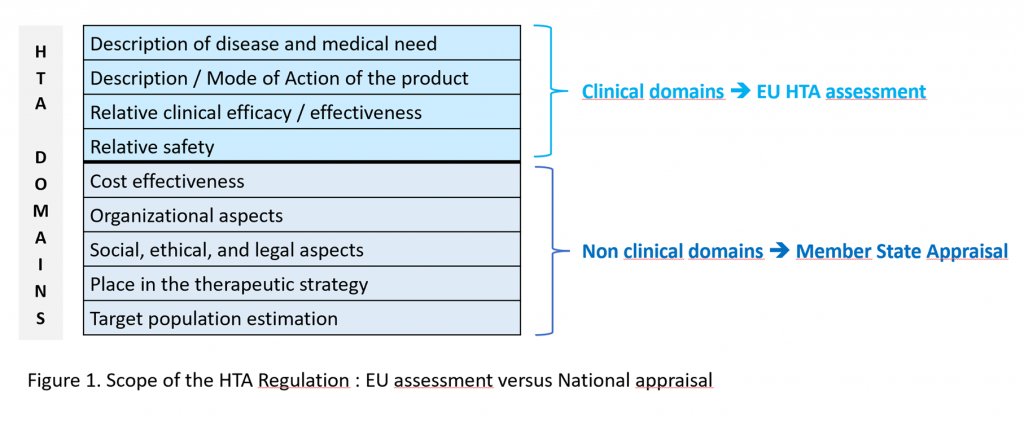
The HTA Regulation focuses on clinical aspects of HTA, i.e., the relative clinical effectiveness and relative clinical safety of a new health technology compared with existing technologies. The pillar of the future EU HTA will be the Joint Clinical Assessments (JCAs).
The JCA will take place in two main phases: the scoping phase (defining the assessment team and the PICO) and the assessment phase. The process allows patients and clinical experts to participate at each stage of the process under specific procedures.
Update on the future guidelines
EUnetHTA 21 is currently developing the methodological guidelines and documents of the EU JCA. Some are already published and give the opportunity to anticipate the expectations of the EU HTA assessment regarding some methodological concerns of the development plan.
For some subjects the key question of the acceptability of the methods is still under discussion (i.e. only accept methods that are acceptable to all member states, or include all commonly used methods of evidence).
| Status | Document | Date of publication |
| Published | Scoping process (PICO) | 12-09-2022 |
| In progress | Practical Guideline on Direct and Indirect Comparisons | Q4 2022 – Project plan available |
| Published | Methodological Guideline on Direct and Indirect Comparisons | 29-07-2022 |
| In progress | Endpoints | Q1 2023 – Project plan and draft deliverable available |
| In progress | Applicability of evidence | Q4 2022 – Project plan available |
| In progress | Validity of clinical studies | Q4 2022 – Project plan available |
| Published | Assessment of High-Risk Medical Devices | 29-07-2022 |
| In progress | EUDAMED data reporting template / Guidance for EUDAMED-based TISP process | Q4 2022 |
| In progress | JCA/CA Submission Dossier Template | Q4 2022 – Project plan available |
| In progress | JCA/CA Assessment Report Template | Q4 2022 – Project plan available |
| Published | Procedural guidelines for appointing assessors and co-assessors | 03-06-2022 |
| Published | HTAb technical expert working group | 29-07-2022 |
| In progress | Production of JCA/CA on medical products and medical devices | 2 medical devices (Optilume® and Evoke®) – no medicinal product |
What should health technology developers anticipate?
The scoping, including the PICO’s definition, will be crucial as the first step of the EU HTA process. From now on, health technology developers should consider the available EUnetHTA 21 methodological guidelines in all clinical developments.
To complete the EUnetHTA 21 guidelines, the French HTA body (Transparency Committee of the Haute Autorité de Santé, HAS) will publish a position paper in the BMJ Evidence-Based Medicine in early 2023.
From 2025, the EU HTA process will include the possibility of a Joint Scientific Consultation (JSC) with the EMA to advise health technology developers on the clinical study designs that provide appropriate evidence. Eight JSCs have been planned in EUnetHTA 21 and will take place by 2023. Before 2025, EUnetHTA 21 recommends using the possibilities to interact with national bodies to define the PICO, especially the comparators.
PHAROS as European healthcare consultancy group with highly experienced market access experts in all major European countries has a good knowledge of European processes and offers a global support for early consultations.
Feel free to contact us via email and we will be happy to advise you!
References
European Parliament and the Council of the European Union. Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on health technology assessment and amending Directive 2011/24/EU; 2021. Accessed Dec 12, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32021R2282&from=EN
European Commission. Regulation on Health Technology Assessment. Accessed Dec 12, 2022. https://health.ec.europa.eu/health-technology-assessment/regulation-health-technology-assessment_en
European Commission. Questions and Answers: Adoption of Regulation on Health Technology Assessment. Accessed Dec 12, 2022. https://ec.europa.eu/commission/presscorner/detail/en/qanda_21_6773
European Network for Health Technology Assessment (EUnetHTA). Joint HTA Work. Accessed Dec 12, 2022. https://www.eunethta.eu/jointhtawork/
Conference on the new Regulation on Health Technology Assessment (HTA). 22 June 2022. Accessed Dec 12, 2022. https://health.ec.europa.eu/events/conference-new-regulation-health-technology-assessment-hta-2022-06-22_en