The Joint Clinical Assessment (JCA) and its implication for the industry
The European (EU) Regulation on Health Technology Assessment (HTA) came into force in January 2022 and creates a permanent framework for cooperation between member states in JCA regarding certain new therapies. This article aims to provide an overview of the current understanding time frame of implementation and some implications for the JCA industry.
What does the JCAs cover?
Recital 14 of the regulation states: “…, the joint clinical assessments …. constitute a scientific analysis of the relative effects of the health technology as assessed on the health outcomes against the chosen parameters which are based on the assessment scope. The scientific analysis will further include consideration of the degree of certainty of the relative effects, taking into account the strengths and limitations of the available evidence.”

Joint clinical reports are meant to replace national assessments with respect to the clinical part of HTAs and should form the basis of national decision-making in all EU countries. However, national HTA bodies have considerable discretion to deviate from the JCA report as they require new data and evidence as well as new comparators. JCA reports are not legally binding. Since the national HTA bodies will be included in the EU debate, the acceptance of JCA at the national level is expected to be high.
According to the founding principles of the EU, the organization of health services, allocation of resources and reimbursement, as well as pricing decisions remain under the sovereignty of the member states. Therefore, JCA covers only the clinical assessment. It is important to note that the JCA at the EU level is strictly separated from value assessment and price negotiations. It does not include any recommendations on a product’s value or its reimbursement. However, the choice of the comparator and the endpoints will be made in the future at the level of the EU, which is very important for price negotiations.
Joint Assessment Process
Like the scientific evaluation of marketing authorization applications by the European Medicines Agency (EMA), the JCA will be conducted by an assessor and co-assessor. These assessors will be from different member states and will be responsible for making the clinical assessment, drafting a report, and consulting with stakeholders. The Coordination Group will then approve the report, which will then be published by the European Commission (EC). The timing of JCA for medicinal products will be linked to the central marketing authorization procedure that will ensure its timeliness in supporting member states’ decision-making at the time of launch.
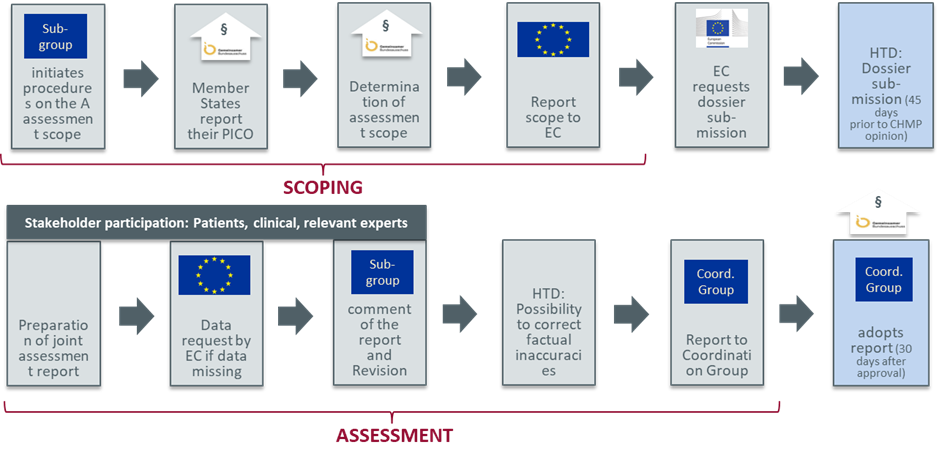
CHMP: Committee for Medicinal Products for Human Use; EC: European Commission; HTD: Health Technology Developer; PICO: Population, Intervention, Comparators and Outcomes
The value of initial engagements (EUnetHTA 21)
Companies affected by the new regulation while the clinical studies are still in the planning phase may request Joint Scientific Consultations (JSC) from the Member State Coordination Group. The consultation topics may include the clinical trial design, choice of comparator, endpoints, interventions, health outcomes, and patient populations.
The developmental guidance of documentation methodology on JCA, JSC and transversal activities that is to be adopted by the Coordination Group from 2021 to 2023 was endorsed by the European Network for HTA (EUnetHTA21). The draft guidance documents developed under EUnetHTA 21 will be subjected to public consultations from May 2022. Unfortunately, due to limited resources, only some JSC can be done each year by the Coordination Group. This means that not all affected companies will be able to seek scientific advice which will prove as a challenge for these companies.
How will the EU HTA regulation work?
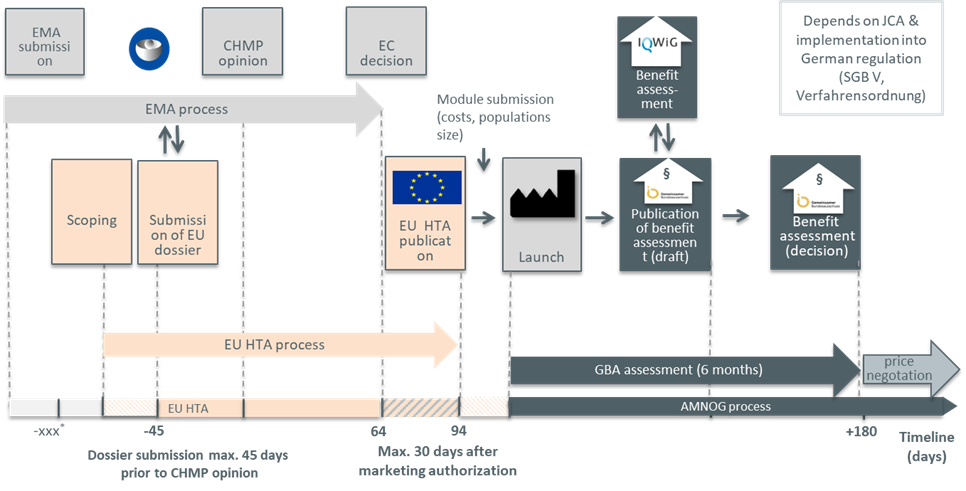
*not yet determined
AMNOG: Gesetz zur Neuordnung des Arzneimittelmarktes; CHMP: Commitee for Medicinal Products for Human Use; EC: European Commission; EMA: European Medicines Agency; IQWIG: Institute for Quality and Efficiency in Health Care
Outlook
The harmonized procedure can lead to a duplication of work at the national level, with follow-on assessments or more evidence that is requested by the Member States. To prevent low uptake of EU HTA reports and increase usability to many member states, an agreement must be reached on the scope and data requirements of the harmonized procedure.
Divergent issues of the HTA processes and methodologies remain to be solved. EUnetHTA21 currently involves all member states’ of HTA bodies in the process of methodological guidance that should be used for JCA. There must be an agreement between the national HTA bodies for particular comparators, endpoints, and the use of data other than those generated in RCTs.
There is still a lot of uncertainty about the resources for JSC. Due to limited resources, JSC may only be made available to some developers.
The success of the EU HTA Regulation to improve patient access to innovative health technologies across Europe is based on the detailed methodology and procedural regulations currently being developed. Manufacturers should therefore engage in the public consultations that will be held by EUnetHTA21 throughout this year in order to present the industry’s perspective on the JCA methodology that will form the basis for the future Pan-European joint HTA.
Any questions?
SmartStep Consulting has been intensively accompanying the development process of the EU HTA procedure since its start in 2018. Due to our extensive experience in the national benefit assessment process and memberships in several European partner associations (PHAROS, EUCOPE), we are well positioned, strongly networked and your reliable partner in the areas of the market access strategy, preparation of dossiers, and early consultation. This concerns AMNOG procedures as well as future EU projects.
Please feel free to contact us via email and we will be happy to advise you!
References
European Parliament and the Council of the European Union. Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on health technology assessment and amending Directive 2011/24/EU; 2021. Accessed July 04, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32021R2282&from=EN
European Commission. Regulation on Health Technology Assessment. Accessed July 04, 2022. https://health.ec.europa.eu/health-technology-assessment/regulation-health-technology-assessment_en
European Commission. Questions and Answers: Adoption of Regulation on Health Technology Assessment. Accessed July 04, 2022. https://ec.europa.eu/commission/presscorner/detail/en/qanda_21_6773