The long road to EU HTA: An update to the current status of implementation
The issues at hand
On 13 December 2021 the EU Regulation on HTA (Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on health technology assessment and amending Directive 2011/24/EU) was adopted. Almost four years after the European Commission (EC)’s proposal in January 2018 the ordinary legislative procedure has been completed. After successful interinstitutional negotiations with both colegislators, the Council of the EU formally adopted its first reading position on 9 November 2021, and the European Parliament closed its early second reading position on 13 December 2021.
The political goal for this EU HTA regulation is that it contributes to improving the availability for EU patients of innovative technologies, such as medicines and certain medical devices. This regulation will also reduce duplication of work and harmonize the methodologies used for the clinical aspects of the evaluation. The regulation replaces the current system based on the voluntary network of national authorities (HTA network) and the EU-funded project-based cooperation (Joint Action EUnetHTA) with a permanent framework for joint work.
From 12 January 2025, JCAs will apply for medicinal products with new active substances for oncological indications and advanced therapy medicinal products (ATMPs). From 13 January 2028 onwards, orphan drug manufacturers must submit a dossier for the JCA. From 13 January 2030, all other medicines authorized under the EU centralized procedure will be subject to JCAs. After this date, any application for marketing authorization of these products submitted to the European Medicines Agency (EMA) will require a JCA dossier.
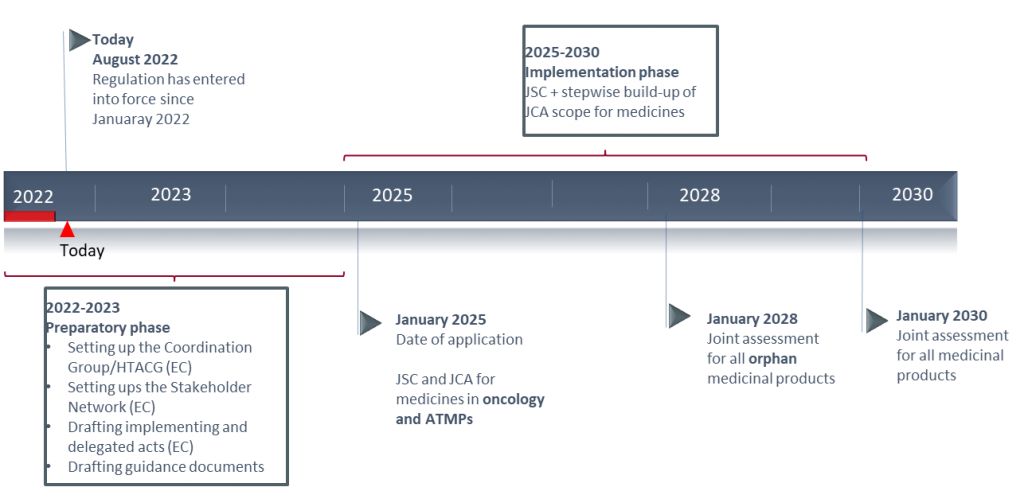
ATMP: Advanced therapy medicinal products; EC: European Commission; HTACG: Health Technology Assessment Coordination Group; JCA: Joint Clinical Assessment; JSC: Joint Scientific Consultation
JCA: The central pillar of the new EU HTA
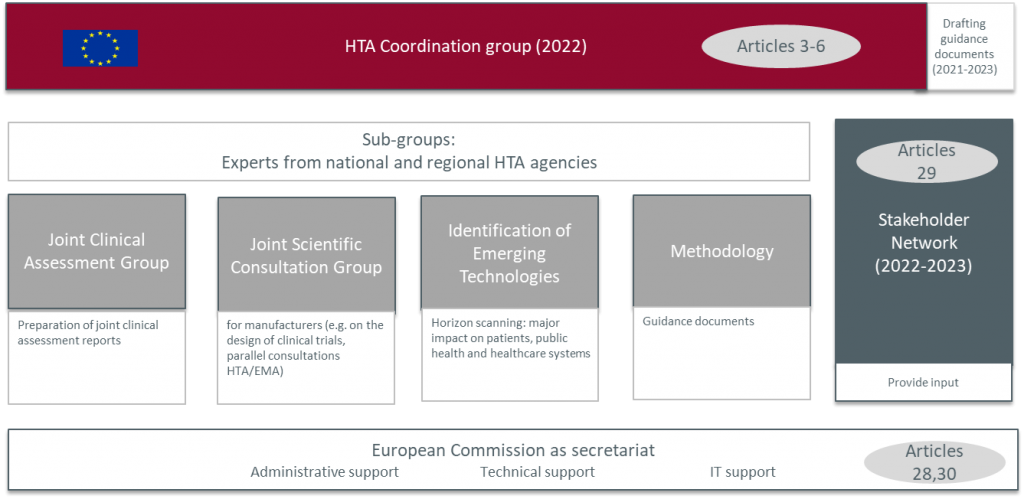
The JCA will be organized and coordinated by the “Member State Coordination Group on HTA” (Coordination Group) is made up of representatives from national HTA bodies and is responsible for overseeing the collaboration of the JCA’s and designated national experts in sub-groups dedicated to specific types of joint work:
- JCA – mandatory assessments focusing on the most innovative health technologies
- JSC – offering manufacturers advice on evidence requirements for the HTA submission
- Identification of emerging health technologies
- Voluntary cooperation on areas outside of the mandated scope
External experts, including clinicians and patients, will also be able to provide input during the preparation of joint work (JCA and JSC). Instead, stakeholder organizations, including patient organizations, healthcare professional organizations, clinical and learned societies, as well as health technology developers and payers will be able to provide input on non-product related issues such as on the drafting and preparation of methodological guidance documents.
Are you interested in more information about EU HTA? Soon you will receive more details about the EU Process. In the meantime, you are welcome to contact us by email and we will be happy to advise you more in detail!
References
European Parliament and the Council of the European Union. Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on health technology assessment and amending Directive 2011/24/EU; 2021. Accessed July 04, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32021R2282&from=EN
European Commission. Regulation on Health Technology Assessment. Accessed July 04, 2022. https://health.ec.europa.eu/health-technology-assessment/regulation-health-technology-assessment_en
European Commission. Questions and Answers: Adoption of Regulation on Health Technology Assessment. Accessed July 04, ’22.https://ec.europa.eu/commission/presscorner/detail/en/qanda_21_6773